چربی بر مای لایف
محلول چربی بر مای لایف کنسانتره ویژه شستشو و پاکسازی آلودگی های پروتئینی مای لایف یک محلول شوینده کنسانتره با کاربرد عمومی برای شستشو و پاکسازی انواع سطوح می باشد.
مزایا :
کنسانتره با قیمت مقرون به صرفه ، بوی مطبوع ، فاقد اثر خورندگی و رنگ بری
دارای قدرت لکه بری بالا ، متعادل کردن PH سطوح محیطی

Solution MY LIF
برای مشاهده محصولات دیگر برروی کلمه ی“ورودبه سایت“ کلیک نمایید .
موارد مصرف محلول چربی بر مای لایف :
در غلظت ۱ تا ۳ درصد :
پاکسازی سطوح آغشته به خون و ترشحات پس از بی خطرسازی خون پاکسازی عمومی سطوح کف دیوار ، اثاثیه ، شیشه ها
در غلظت ۵ تا ۱۰ درصد :
شستشو و پاکسازی چربی ها و آلودگی ارگانیک روی سطوح بسیار کثیف مانند هود آشپزخانه
دستور مصرف :
۱ . برای پاکسازی عمومی سطوح مقدار ۱۰ تا ۲۰ سی سی از کنسانتره را در آب معمولی حل کرده و با آبفشان روی سطوح را آغشته کرده و با دستمال تمییز پاکسازی نمایید. (یا به روش دستمال مرطوب)
۲ . برای شستشوی سطوح بسیار کثیف مقدار ۵ تا ۱۰ سی سی از محلول را در آب ترجیحا گرم حل کرده و سطوح را آغشته کنید. چند دقیقه برای تاثیرگزاری زمان بدهید تا الودگی ها نرم و سست شوند ، سپس محل را تمییز نمایید.
شرایط نگهداری و ایمنی محلول چربی بر مای لایف :
در محیط خشک و دمای ۱۵+ تا ۳۵+ درجه نگهداری شود.
پس از اسنتشاق :
بو و بخارات خاصی ندارد. جریان طبیعی هوا برقرار باشد.
در صورت تماس با چشم :
با مقادیر فراوان از آب شستشو شود.
در صورت بلعیدن :
فورا مراقبت های پزشکی درخواست شود.
Introduction
Pathogenic organisms can be introduced into a veterinary clinic or animal housing facility
through a variety of ways.
For this reason, biological risk management (BRM) protocols are necessary to prevent, contain and eliminate the spread of disease.
Disinfection protocols, when
implemented correctly, can be a cost-effective means of reducing pathogenic organisms and are
an important step in any biological risk management program.
Prevention of disease is typically easier and more cost-effective than addressing an outbreak situation.
Therefore, development and implementation of a step-by-step disinfection protocol for the control and prevention of infectious disease has become essential for farms and clinics.
Disinfection protocols may vary depending on the need of the farm or clinic. No single disinfectant is adequate for all situations.
Disinfection protocols used on a daily basis will differ from those needed to control an infectious disease outbreak.
However, both have one component in common; thorough cleaning and washing prior to the application of any disinfectant is essential.
The purpose of this handout is to provide 1) an overview of factors to consider when developing and implementing an effective disinfection protocol, 2) an overview of chemicals used for disinfection, their advantages and limitations, and 3) essential steps of an effective disinfection protocol.
Following development of a disinfection plan, it is equally important to train personnel of the proper procedures to use and safety issues involved as well as to have the steps posted in prominent locations throughout the facility to serve as a reminder of proper disinfecting techniques.
محلول چربی بر مای لایف
Disinfectants Defined
Disinfecting agents are registered by the Environmental Protection Agency (EPA) as
“antimicrobial pesticides” and are substances used to control, prevent, or destroy harmful
microorganisms (i.e., bacteria, viruses, or fungi) on inanimate objects and surfaces.
These
antimicrobial products have traditionally included sanitizers, disinfectants, and sterilants.
Data
on a product’s chemistry, efficacy, toxicity to humans, animals and plants, and other
parameters must be tested and submitted to the EPA prior to the marketing of the chemical.1
Chemical disinfectants can have various effects against microorganisms.
Therefore, a basic understanding of the different chemical agents is important.
Biocide or germicide refers to chemical agents that kill microorganisms.
These general terms includes disinfectants, antiseptics and antibiotics.
Germicides and biocides generally react with proteins, specifically essential enzymes of microorganisms.
Actions may include oxidation, hydrolysis, denaturation or substitution.
2 When a killing action is implied, the suffix –cide (e.g. biocide, bactericide, virucide, sporicide) is used, while –static (e.g. bacteriostatic, virostatic, sporostatic) is added when an organism’s growth is merely inhibited or it is prevented from multiplying.
3 Sanitizers do not destroy or eliminate all bacteria or microorganisms, but reduce the number of microbial contamination on inanimate surfaces to levels that are considered safe from a public health standpoint.
Many sanitizers are a formulation of a detergent and disinfectant.
Surface Quick سورفیس کوئیک
Disinfection 101
Disinfectant describes a product applied directly to an inanimate object.
It destroys or irreversibly inactivates most pathogenic microorganisms, some viruses, but not usually
spores.
2,4,5,6 In comparison, antiseptics are applied to the surface of living organisms or tissues to prevent or stop the growth of microorganisms by inhibiting the organism or by destroying them.2
Sterilization refers to the process, either physical (i.e., extreme heat) or chemical (i.e.
ethylene oxide), that destroys or eliminates all forms of life, especially microorganisms.2,4,6
Detergents serve to disperse and remove soil and organic material from surfaces allowing a
disinfectant to reach and destroy microbes within or beneath the dirt.
These products also reduce surface tension and increase the penetrating ability of water, thereby allowing more organic matter to be removed from surfaces.
2 Some disinfectants have detergent properties (i.e., chlorine compounds, iodophors, QACs).
محلول چربی بر مای لایف
Detergents are classified in three categories:
cationic, anionic and non-ionic. Cationic detergents
are positively charged solutions, and with the exception or quaternary ammonium compounds
(QACs), are seldom used a cleaning ingredients.
Anionic detergents, or soaps, are negatively
charged alkaline salts of fatty acids.
They are less ideal for cleaning because they can be
excessively foamy, creating a residue that may allow soil and microorganisms to accumulate.
Nonionic (uncharged) detergents are very good emulsifiers, have good penetration and
dispersion, are effective at lowering surface tension, and have reduced foaming properties.
These products do not typically complex with metallic ions, such as those found in hard water.
Most commercial detergents are a combination of anionic and non-ionic.
محلول چربی بر مای لایف
Disinfectant Labels7
Product labels contain important information on the proper use and hazards of a chemical.
This
information may often be overlooked however it is a violation of federal law to use a product in a manner inconsistent with its labeling.
Therefore, strict attention must be given to the proper use of a product with regard to its pplication, effectiveness, and associated hazards (human, animal, and environment).
This information will assist in decisions for infection control efforts.
محلول چربی بر مای لایف
Label Claims
Disinfectants may have a range of uses and label claims, such as cleaner, deodorizer, sanitizer,
disinfectant, fungicide, virucide or ‘for hospital, institutional and industrial use’.
Label claims are
primarily determined by three test microorganisms, Staphylococcus aureus, Salmonella cholerasuis, and Pseudomonas aeruginosa.
• Limited efficacy is a claim of disinfection or germicidal activity against one specific microorganism group (eg. Gram-negative or Gram-positive).
Gram-positive
designation comes from effectiveness against Staphylococcus aureus, while Gramnegative bacteria claims must be effective against Salmonella cholerasuis.
The label must specify the group against which the product is effective.
• General-purpose or broad-spectrum is a claim of effectiveness against Grampositive and Gram-negative bacteria.
This claim must be supported by efficacy testing against Staphylococcus aureus and Salmonella cholerasuis.
Disinfection 101
• Hospital or medical environment claim must be supported by efficacy testing against S. aureus and S. cholerasuis but also efficacy against the nosocomial bacterial pathogen, Pseudomonas aeruginosa.
• Claims against pathogenic fungi or other microorganisms are permitted, but not required, on the label following standardized testing procedures.





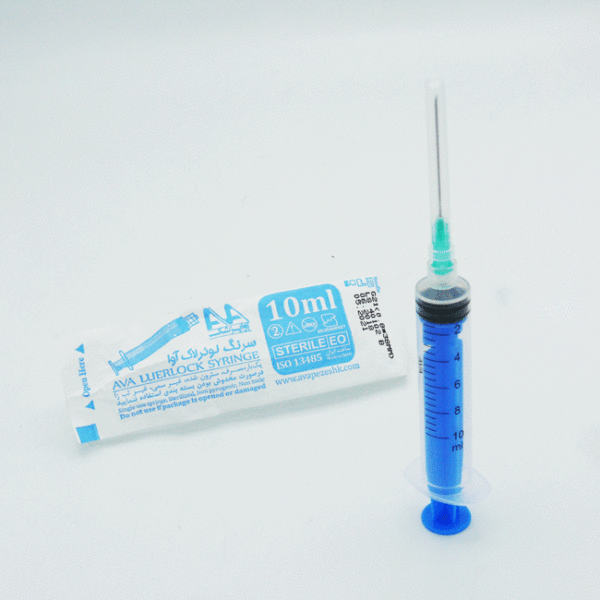











نقد و بررسیها
هنوز بررسیای ثبت نشده است.